Responsible Research
ISTA is committed to the highest academic standards in research ethics and integrity.
ISTA is committed to the highest international academic standards of responsible conduct of research. These include Research Integrity and Research Ethics.
Research Integrity focuses on “how you do your research”. Research Integrity addresses aspects related to ensuring the quality, reliability, and honesty of scientific knowledge.
Research Ethics focuses on “what you do in your research”. Research Ethics addresses questions related to research content, topics, and scientific approaches.
It is the responsibility of each employee of ISTA to foster an environment that promotes Research Integrity and Ethics, and that does not tolerate misconduct.
Research Integrity (Good Scientific Practice) focuses on ensuring the principles of quality, reliability, honesty, and accountability of scientific knowledge.
Guidelines for Good Scientific Practice
All persons involved in research at the Institute of Science and Technology Austria (ISTA) are obliged to adhere to the standards of Good Scientific Practice.
At ISTA, the Guidelines for Good Scientific Practice of the Austrian Agency for Research Integrity (Österreichische Agentur für wissenschaftliche Integrität, OeAWI) are in place. They define the principles of integrity in research and scholarship and provide definitions of good scientific practice and of scientific misconduct.
Another important document is the European Code of Conduct for Research Integrity published by the ALLEA (All European Academies). The European Code of Conduct for Research Integrity is part of the contractual grant agreement of all EU research projects in the EU framework program Horizon Europe.
Responsible Use of Generative AI in Research
Generative AI tools have become powerful allies for researchers, enhancing scientific and academic output across disciplines. At ISTA, we are committed to ensuring that generative AI tools are used in a responsible and acceptable manner.
The Responsible Use of Generative AI in Research guide at ISTA outlines key practical dos and don’ts for scientists to follow when using generative AI in research and academic activities, as summarized in the image below.
Furthermore, our comprehensive internal document provides additional details and emphasizes the importance of responsibility, transparency, research ethics, data protection, and privacy, with a strong focus on maintaining scientific integrity and accountability throughout these topics.
The recommendations aim to promote a balanced approach that maximizes the benefits of AI innovations while mitigating potential risks and adverse impacts. In addition, the guidelines emphasize the need for continued education and awareness of the evolving landscape of AI technologies. The scope of the use of generative AI tools in research is a rapidly evolving topic. At ISTA, the guidelines for scientists are aligned with current international standards. Specifically, they follow the recommendations stated in the Living Guidelines on the Responsible Use of Generative AI in Research from the European Commission for incorporating generative AI into their research.
Scientific record keeping and data management
Every person performing experimental work at ISTA has to keep records of the conducted experiments. For scientific record keeping, either a paper lab notebook or an electronic lab notebook can be used.
A well-kept lab notebook provides a reliable reference for writing up methods and results of a study, for preparation of formal reports, papers, and presentations. A comprehensive notebook permits one to reproduce any part of a methodology completely and accurately. It can be an invaluable source of information for a variety of purposes, including demonstration of adherence to standards of good scientific practice and of academic and ethical integrity. In addition, it is a record for determination of legal claims of discovery where new inventions are concerned.
More information about research data management, Open Access, the institutional repository ISTA Research Explorer, and related topics can be found on ISTA’s library website.
Ombudspersons for Good Scientific Practice at ISTA:
Two Ombudspersons are available at ISTA for providing advice and information regarding research integrity to all employees of ISTA, and for receiving complaints and allegations of research misconduct.
The Ombudspersons are independent in their work as contact persons for confidential meetings.
The Ombudspersons should be addressed if there are any complaints regarding scientific or academic behavior. In particular, they are the persons of contact for scientists who have questions regarding or wish to report a possible incidence of misconduct related to research, teaching, and/or scientific supervision at ISTA.
Ombudspersons at ISTA:
Beatriz Vicoso (beatriz.vicoso@ista.ac.at)
Verena Seiboth (verena.seiboth@ista.ac.at)
To ensure fair and transparent handling of inquiries to Ombudspersons and allegations research misconduct, ISTA has detailed rules in place that define the confidentiality, information and reporting workflow and handling of allegations at the different steps of an investigation procedure. An overview can be found here.
For conflicts that do not involve aspects related to research integrity, the Conflict Management System of ISTA provides options for handling confidential issues and conflict situations.
Every scientist should take ethical principles and responsible approaches to research into account and should ensure that their work is in agreement with such principles. Researchers thus have a professional responsibility to critically reflect on potential ethical limitations of their research. In addition to the obligation of scientists to adhere to legal provisions, a responsible approach towards research involves avoiding or minimizing the risks of harm to humans and the environment and potential misuse (dual use) of their research results.
Ethics screening processes at ISTA are aligned to the requirements and definitions for the ethics topics that are defined for EU research projects in the framework program Horizon Europe.
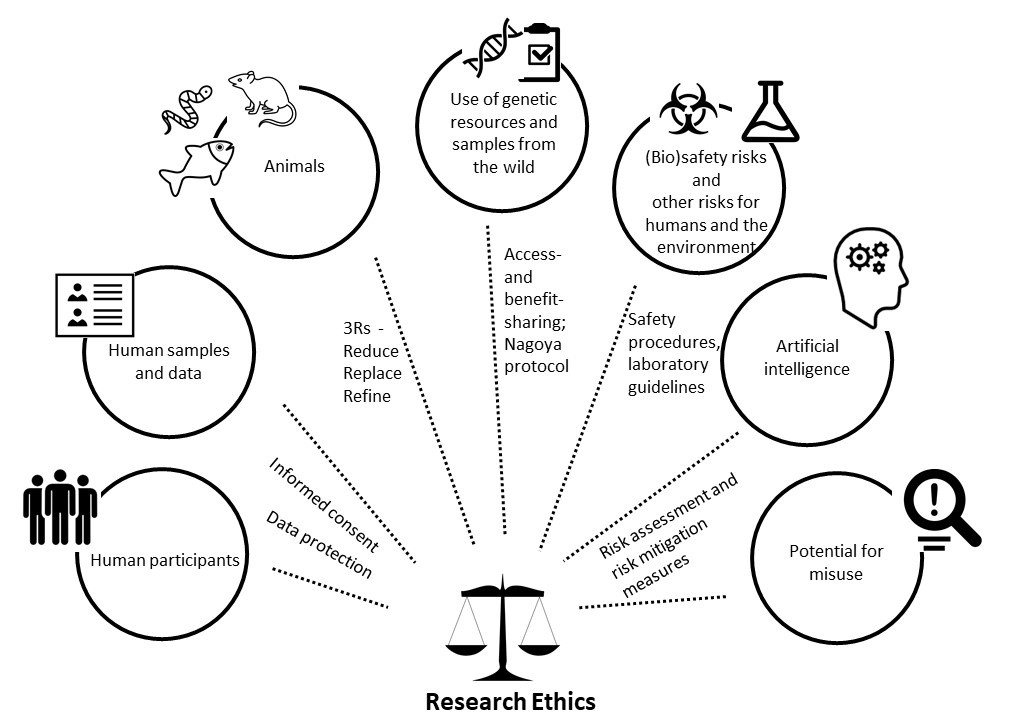
The Ethics Guidance for EU grants from the European Commission (“EU Grants – How to complete your ethics self-assessment”) provides a comprehensive overview of these ethics topics.
ISTA Ethics Officer: Krishna Mutanwad (krishna.mutanwad@ista.ac.at) or ethicsofficer@ist.ac.at
In order to ensure the maintenance of high ethical research standards at ISTA, the Ethics Officer and the Ethics Committee can be consulted on questions regarding ethical issues of research projects at ISTA.
ISTA Ethics Committee:
The Ethics Committee can be consulted on ethical issues regarding ongoing and planned research projects. If addressed with an inquiry, the Ethics Committee evaluates potential ethical risks of planned and ongoing research projects and scientific approaches, and issues a recommendation if the respective research projects should be performed as outlined in the project description. It does not, however, evaluate topics regulated by law in Austria. In those cases, permission from the respective governmental authorities needs to be obtained.
For questions or inquiries to the ISTA Ethics Committee, please contact the Ethics Officer.
